|
Template:Nucleosynthesis periodic table.Scale by 10x to improve rendering of text. The atomic number of each element increases by one, reading from left to right. Period A horizontal row in the periodic table. Strategy: Locate the elements in the periodic table. Members of a group typically have similar properties and electron configurations in their outer shell. Asked for: order by increasing electronegativity and classification. Formulas indicate the type of compounds formed by each group, with R standing for any element and superscripts. The column headings Reihen and Gruppe are German for row and group. Hydrogen is a non-metal, but it is often put in the middle. 
Just put a URL to it here and well apply it, in the order you have them, before the CSS in the Pen itself. On the basis of their positions in the periodic table, arrange Cl, Se, Si, and Sr in order of increasing electronegativity and classify each as a metal, a nonmetal, or a metalloid. 1 Mendeleev’s Periodic Table, as Published in the German Journal Annalen der Chemie und Pharmacie in 1872. The zigzag, staircase, line in the diagram of the periodic table above separates the metals, on the left, from non-metals, on the right. Update table with more up-to-date data from Jennifer Johnson as discussed on You can apply CSS to your Pen from any stylesheet on the web. Elements in each group have similar properties with other elements in that. Update heavy elements to match, and add elements to lawrencium, key for synthetic elements, and textures to help the colour blind, as per. 5 Groups of the PTE (cont.) Chemical properties would include how reactive they are. GFDL GNU Free Documentation License true trueĬlick on a date/time to view the file as it appeared at that time. These elements are very reactive, and usually occur in nature already combined with. A copy of the license is included in the section entitled GNU Free Documentation License. You may also use the color-coded periodic table chart with names, symbols. Groups 1, 2, and 1318 are the main group elements, listed as A in older. 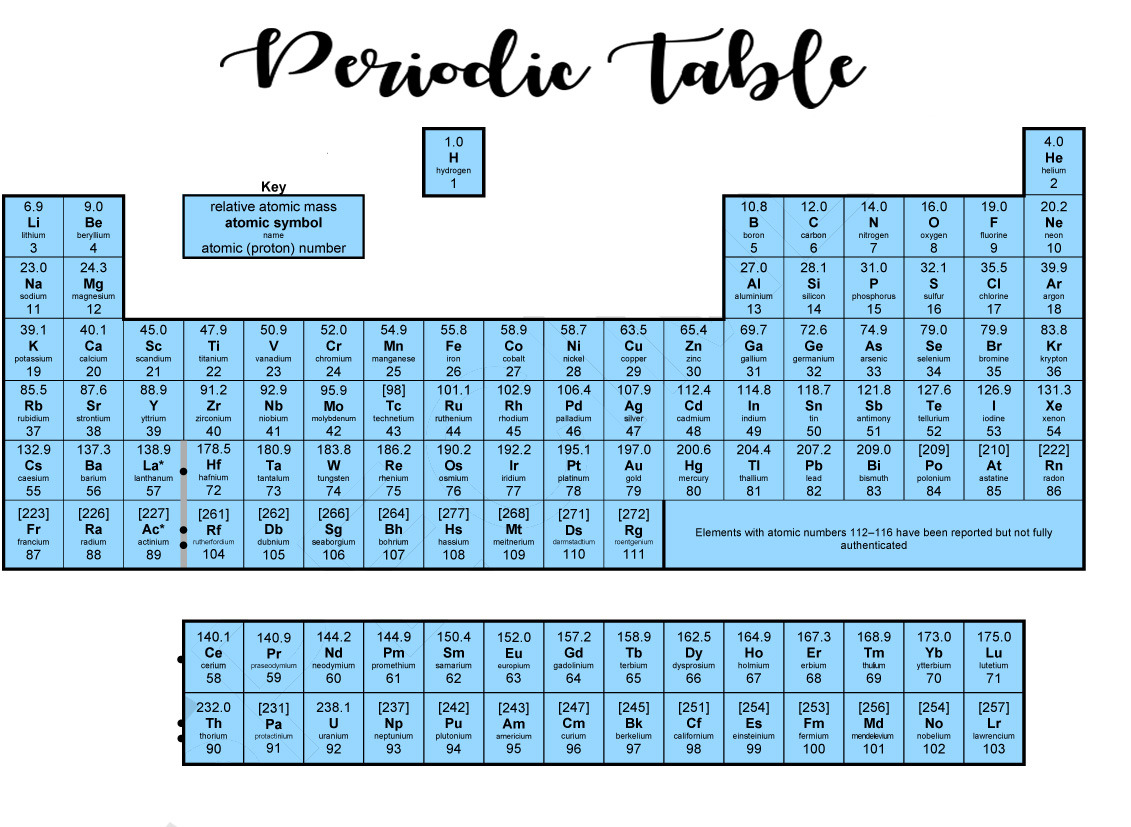
The elements are stacked in such a way that elements with similar chemical properties form vertical columns, called groups, numbered from 1 to 18 (older periodic tables use a system based on roman numerals). Examples: The row starting with hydrogen is 1. The rows are called periods, and they are numbered from 1 to 7. Part 4: Above groups 1, 17, and 18, label the groups reactivity (very reactive. How to Identify It: Period numbers are located on the left-hand side of the table. Permission is granted to copy, distribute and/or modify this document under the terms of the GNU Free Documentation License, Version 1.2 or any later version published by the Free Software Foundation with no Invariant Sections, no Front-Cover Texts, and no Back-Cover Texts. The period indicates the highest energy level attained by electrons of an atom of the element in the ground state. CC BY-SA 3.0 Creative Commons Attribution-Share Alike 3.0 true true These individual element summary pages contain a lot of additional. When exploring the table or list views on this page, please note the links to dedicated pages for each element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
